How Did Clausius Define Entropy?
Clausius, one of the main founders of the modern thermodynamics, is the originator of the term “entropy," but before this coinage he used an expression “equivalence value of transformations" instead. The term “entropy," which is now used as a physical term, was originally economically conceived as equivalent value of exchange. Thermodynamics itself was a field of physics that was economically motivated to improve the thermal efficiency of heat engines. So, it was half physics and half economics. Apart from Clausius’s interest, the term has now become an important keyword for solving the environment and resource problems.

Contents
1. The starting point of Clausius’s theory of heat
The thermodynamics of Clausius was gradually formulated in his papers of 1850, 1854, 1862 and 1865. Among them, the paper in 1850, On the Moving Force of Heat, and the Laws regarding the Nature of Heat itself which are deducible therefrom (Über die bewegende Kraft der Wärme: und die Gesetze, welche sich daraus für die Wärmelehre selbst ableiten lassen) is the starting point of his mechanical theory of heat. I will examine the range and limit of his theory that was already revealed at this early stage.
1.1. The reconciliation between Carnot and Joule
Along with William Thomson (1st Baron Kelvin; 1824 – 1907), Rudolf Julius Emmanuel Clausius (1822 – 1888) is one of the main founders of the modern thermodynamics who succeeded to Nicolas Léonard Sadi Carnot, the pioneer of this science. Unlike Thomson, however, Clausius did not think Joule’s theory that heat can be converted into work contradicted Carnot’s theory that presupposed the conservation of heat. In his paper of 1850, Clausius abandoned the principle of the conservation of heat and accepted Joule’s law as the first law of thermodynamics, while he also accepted Carnot’s principle as the second law of thermodynamics.
On a nearer view of the case, we find that the new theory is opposed not to the real fundamental principle of Carnot, but to the additional principle “no heat is lost," for it is quite possible that in the production of work both may take place at the same time; a certain portion of heat may be consumed,and a further portion transmitted from a warm body to a cold one; and both portions may stand in a certain definite relation to the quantity of work produced.[1]
The “real fundamental principle of Carnot" is such that the production of motive power is due to the transportation of heat from a warm body to a cold body[2]. After stating this principle in his treatise, Reflections on the Motive Power of Fire, published in 1824, he gave up the principle of the conservation of heat and admitted in his posthumous manuscript that part of heat could be converted into work[3]. Clausius gained the final insight of Carnot into the reconciliation between two principles without reading the manuscript 18 years after his death and thus succeeded in discovering the second law of thermodynamics prior to Thomson[4].
1.2. The concept of work in thermodynamics
According to Carnot and Clausius, a heat engine requires another condition to produce motive power besides a warm body and a cold body, namely what Carnot called agent, usually the water vapor, which is in contact with a warm body and a cold body alternately.
As a transmission of heat may take place by conduction without producing any mechanical effect when a warm body is in contact with a cold one, if we wish to obtain the greatest possible amount of work from the passage of heat between two bodies, say of the temperatures t and τ, the matter must be so arranged that two substances of different temperatures shall never come in contact with each other.[5]
Strictly speaking, the third body between a warm body and a cold body is necessary only when heat engines must produce the repetitious and periodic work. If the non-repetitious and non-periodic work suffices, we can make two substances of different temperatures come in contact with each other. The direct contact results in the expansion of the cold body, which can be regarded as the result of work.
Although Clausius inquired into the non-repetitious and non-periodic work later, he confined himself to the conversion of heat to work in the Carnot cycle in his paper of 1850. Carnot chose the principle of steam engines as the subject of his study, because it was the cutting-edge technology at that time and a matter of great interest, but Clausius had another reason for starting his thermodynamics from the analysis of the Carnot cycle: it was still unknown in those days where the heat is gone that is not converted to work and the Carnot cycle independent of this problem could simplify the model of thermodynamics.
Though he developed a kinetic hypothesis of gases and thought that the unused heat would change the interior molecular state of gas, he avoided details in his paper of 1850. He assigned a symbol U to the energy that a gas has in it and formulated a differential equation that represents where heat goes[6].
According to Clausius, the symbol U “comprises the supervening free heat and the heat used for interior work, if such be present[7]." It means he assumed “interior work" as well as exterior work that we usually call just work. He explained the difference of two works as follows.
The work produced is of a twofold nature. In the first place, a certain quantity of work is necessary to overcome the mutual attraction of the particles, and to separate them to the distance which they occupy in a state of vapour. Secondly, the vapour during its development must, in order to procure room for itself, force back an outer pressure. We shall name the former of these interior work, and the latter exterior work, and shall distribute the latent heat also under the same two heads.[8]
Latent heat is the energy necessary for a phase transition, for example, the melting of ice or the boiling of water. It does interior work that cuts off intermolecular forces. If the interior work is work, why shouldn’t “the supervening free heat" that increases the kinetic energy of molecular motion be treated as work?
Physics defines work as the scalar product of the force and displacement vectors. Thermodynamics confines the term only to the work done for a macroscopic object such as a piston, but it is theoretically possible to apply this term to the scalar product of the microscopic force and displacement vectors resulting from the change in the microscopic kinetic energy and the microscopic potential energy of water molecules. Thermodynamics does not call it work, because such microscopic work is not useful for users of heat engines.
In thermodynamics, the internal energy is the energy needed to create the system but excludes the energy to displace the system’s surroundings, the kinetic energy of motion of the system as a whole, and the potential energy of the system as a whole due to external force fields. Though it is a macroscopic quantity, internal energy can be explained in microscopic terms by two components. One is the microscopic kinetic energy due to the microscopic motion of the system’s particles (translations, rotations, vibrations). The other is the potential energy associated with the microscopic forces, including the chemical bonds, between the particles, and with the static rest mass energy of the constituents of matter.
Today we call U the internal energy and we know that its increase is the increase of the kinetic and/or potential energy of the particles that constitute the system. Clausius was the pioneer of kinetic theory of gases and he knew what energy U is. But it was just an unverified hypothesis and this is why he said “if such be present" in the quoted passage and assigned the symbol U meaning “unknown (unbekannt in German)" to the internal energy. If he had founded his theory on a mere hypothesis, his whole theory would become doubtful. Clausius studied the conversion of heat to exterior work in the Carnot cycle to avoid such a doubt.
When any body changes its volume, the change is always produced or expended by a mechanical work. In most cases, however, it is impossible to determine this with accuracy, because an unknown interior work usually goes on at the same time with the exterior. To avoid this difficulty, Carnot adopted the ingenious contrivance before alluded to: he allowed the body to undergo various changes, and finally brought it into its primitive state; hence if by any of the changes interior work was produced, this was sure to be exactly nullified by some other change; and it was certain that the quantity of exterior work which remained over and above was the total quantity of work produced.[9]
Though we still use U as the symbol for the interior energy, the once unknown U is no longer unknown. To apply the original definition of work to the microscopic object, the change in the interior energy can be considered to be the result of work. Thus we can say the entire heat that a system absorbs can be converted to work, whether the system is the cold body of the heat engine or a cylinder which absorbs friction heat or anything else.
The second law of thermodynamics is often formulated as “not all heat can be converted to work [10]" as a result of misunderstanding Thomson’s Principle[11], but this is not correct. We can not only convert all of work to heat, but we can also convert all of heat to work. As heat is microscopic work, we might have to say we just convert the form of work. While the first law of thermodynamics says the amount of work is constant, the second law says the work becomes useless by conversion in an irreversible process.
You might think that it is inappropriate to introduce such a subjective word as usefulness to physics, but here we must recollect that thermodynamics originally had an economic motive to improve the efficiency of heat engines. We must distinguish the physically defined concept of “work" in a wide sense from the economically defined concept of “work" in a narrow sense, namely useful work. It is up to you how you define a term, but what is important for thermodynamics is to illuminate what that degeneration of usefulness is.
1.3. Mathematical formulation of Carnot’s principle
In the paper of 1850, Clausius formulated the Carnot’s principle mathematically that would be important for his definition of entropy. The differential equation that I have already quoted represents the relation of the absorbed heat to the change in the internal energy and the exterior work.
To adopt the current notational convention, Eq. 1 can be reformulated this way.
Here δ instead of d is used to indicate the variable is not necessarily the infinitesimal. That is to say, even if the change in the absorbed heat and the exterior work themselves are not infinitesimal, their difference, the change in the interior energy U, can be infinitesimal.
Some other differences from the current notation need to be explained. As the unit of Q was not Joule but calorie at that time, the constant A, the equivalent of heat for the unit of work, was necessary for the unit conversion. The unit of t was Celsius temperature and added a=273 to be the absolute temperature. R was not the same as the current gas constant. It was therefore “different from different gases, being inversely proportional to the specific weight of each[12]." To make R a constant, we should multiply R and the amount of substance, denoting nR."
In the isothermal change, dU=0 and the equation is as follows.
Integrated with volume (v), the equation below[13] is derived (subscript 0 denotes an initial value).
This equation proves the law of Carnot: “When a gas varies in volume without change of temperature, the quantities of heat absorbed or liberated by this gas are in arithmetical progression, if the increments or the decrements of volume are found to be in geometrical progression[14]."
To adopt the current notational convention, Eq. 4 can be reformulated this way.
This equation is just a few steps away from Boltzmann’s entropy formula. Divided by the absolute temperature T, this equation
is equivalent to the equation below.
Here N stands for the number of moleculres anb NA the Avogadro constant. Substitute Boltzmann constant k for R/NA, and you get Boltzmann’s entropy formula.
Taking into consideration that he was the pioneer of the kinetic theory of gases, we can safely say his thermodynamics came near to the statistical mechanics.
2. The theorem of the equivalence of transformations
In his paper of 1854 On a modified form of the second fundamental theorem in the mechanical theory of heat (Über eine veränderte Form des zweiten Hauptsatzes der mechanischen Wärmetheorie), Clausius developed the second law of thermodynamics formulated in the paper of 1850 and defined the equivalence value of transformations, which was later renamed entropy.
2.1. What is the equivalence value of transformations?
In this paper Clausius called the Carnot principle “the Theorem of the Equivalence of Transformations (Satz von Aequivalenz der Verwandlungen) and formulated it this way.
In all cases where a quantity of heat is converted into work, and where the body effecting this transformation ultimately returns to its original condition, another quantity of heat must necessarily be transferred from a warmer to a colder body; and the magnitude of the last quantity of heat, in relation to the first, depends only upon the termperatures of the bodies between which heat passes, and not upon the nature of the body effecting the transformation.[15]
Simply put, “heat can never pass from a colder to a warmer body without some other change, connected therewith, occurring at the same time[16]." If you want to convert heat into useful work, you must transfer heat from a hot body to a cold body. Conversely if you want to transfer heat from a cold body to a hot body, you must convert useful work into heat.
Let’s schematize this principle of exchange. As the figure below shows, when we burn fossil fuels and run a steam engine or any thermal power plant, we transform heat into useful work in compensation for narrowing the temperature difference.

In contrast with this, as the figure below shows, when we use an air conditioner or a refrigerator, we widen the temperature difference in compensation for transforming useful work done by electricity into heat.
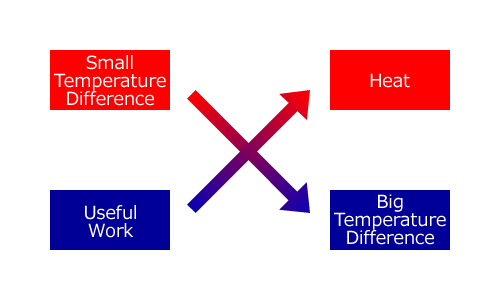
These thermodynamic exchanges remind us of an exchange of economic equivalents: you must pay money for others doing useful work for you and conversely you must do useful work for others to earn money. Just as spending money is easy while earning it is difficult, the transfer of heat from a hot body to a cold body or transforming useful work into heat is easy while the transfer of heat from a cold body to a hot body or transforming heat into useful work is difficult. This simile might help you to understand why Clausius named the law the Theorem of the Equivalence of Transformations.
2.2. Why can’t the equivalence value be negative of itself?
In the economic exchange the price that money established denotes the equivalence. What quantity denotes the equivalence of the exchange in the Carnot principle? Clausius defined
as “the equivalence value of transformations[17]," where T is the absolute temperature and Q is the heat a system absorbs. He found that the value is positive in such cases as the transfer of heat from a hot body to a cold body or transforming useful work into heat and that the value is negative in such cases as the transfer of heat from a cold body to a hot body or transforming heat into useful work. To use the economic analogy, the increase in the equivalence value of transformations corresponds to the increase in expenses for work.
Let’s recognize it. First suppose a quantity of heat Q transfers from a hot body whose absolute temperature is T1 to a cold body whose absolute temperature is T2. Consider Q to be positive when a system absorbs it and negative when a system gives it off. Then the equivalence value of transformations is
By definition, T1>T2. Therefore the value is always positive. Contrariwise it must be negative when heat transfers from a cold body to a hot body. Useful work can be regarded as an unlimitedly hot body. So, the equivalence value of the transformation from useful work into heat is positive.
Contrariwise it must be negative when heat is transformed into useful work. In any case, it cannot be negative as a whole.
When heat a system absorbs changes the temperature of the system, you must divide Q into infinitesimals and integrate them so that Eq. 9 can be written as follows.
The sum of the equivalence value of transformations is zero in a reversible cycle like the Carnot cycle. In such an ideal exchange of work and heat, the work generated from the transfer of heat from a hot body to a cold body can transfer the heat from the cold body to the hot body, thus restoring the original state. This idealized cycle is impossible in real heat engines which cause vortex motions in their cylinders and give off friction heat in operation. Their cycles are irreversible and the equivalence value of transformations is positive.
To use the analogy of the economic exchange, in an idealized market where wages have no difference and any exchange has no extra cost, you can pay others for doing work and withdraw the same amount of money doing the same work, thus restoring the original state. But in reality you must pay extra costs such as those of recruiting, transportation, finance and so on, so that you cannot withdraw your original money completely.
2.3. The thermodynamic limit of Clausius’s approach
We are often deceived into losing money without receiving any equivalent value of work in the market of service. The equivalence value of transformations in the thermodynamic exchange can also increase without work. Clausius named it “an uncompensated transformation[18]" and gave some examples such as the transmission of heat by mere conduction, the production of heat by friction and by an electric current when overcoming the resistance. As we have already recognized, however, we cannot say that no work is done in these cases, because generated heat increases the interior energy and/or expands the volume, which can be regarded as work at microscopic level, although thermodynamics does not treat them as work on account of uselessness.
Isn’t there, then, any cases where we pay money for no work rather than useless work? The last example that Clausius gave, adiabatic free expansion or Joule expansion, is the perfect example of an uncompensated transformation as to an ideal gas. Joule expansion is named after James Prescott Joule, who theorized this expansion in detail in 1845, though Joseph-Louis Gay-Lussac (1778 – 1850) also studied it in 1807. Clausius also recognized it as an important problem to solve.
The figure below is a schematic depiction of Joule expansion. (1) First, a volume of gas is kept in the left side of a thermally isolated container via a partition in the middle with the right side of the container being evacuated. (2) Then the partition is opened and the gas evenly fills the whole container to reach an equilibrium state. This process is irreversible and the state (2) does not go back to the state (1) spontaneously.
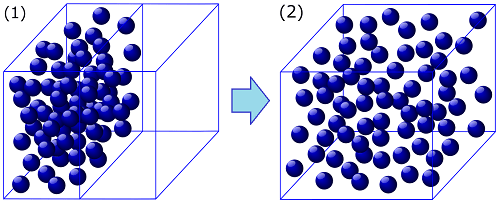
Because the gas that Joule used in his experiment was near an ideal gas, he could not observe a drop in temperature owing to the expansion. Mentioning this irreversible process, however, Clausius avoided details in his paper of 1854, saying “I will not here enter further into the treatment of particular cases[19]."
It could not be helped that Clausius was not able to explain the irreversible process of the Joule expansion in terms of the equivalence value of transformations. The Joule expansion absorbs no heat, does no exterior work and therefore causes no change in the interior energy, that is to say, no change in temperature. As the change in every variable in Clausius’s equation is zero, the increase in the equivalence value of transformations cannot but be zero. But it should be positive, because the process is irreversible. So, the Joule expansion detected the limit of Clausius’s approach, for statistical mechanics instead of thermodynamics is necessary to explain the irreversibility of the Joule expansion.
3. Entropy as the sum of heat and disgregation
In the paper of 1862, “On the Application of the Theorem of the Equivalence of Transformations to Interior Work (Über die Anwendung des Satzes von der Aequivalenz der Verwandlungen auf die innere Arbeit)", and the paper of 1865, “On Several Convenient Forms of the Fundamental Equations of the Mechanical Theory of Heat (Über verschiedene für die Anwendung bequeme Formen der Hauptgleichungen der mechanischen Wärmetheorie), Clausius analyzed the equivalence value of transformations into the transformation-value of heat and the disgregation and proposed a new name, entropy, as their sum.
3.1. The analysis of the interior energy
In his paper of 1862, Clausius confessed that his previous theory was limited in that he confined himself to the consideration of the Carnot cycle.
Since in my former paper I wished to avoid everything that was hypothetical, I entirely excluded the interior work, which I was able to do by confining myself to the consideration of cyclical processes, that is to say, operations in which the modifications which the body undergoes are so arranged that the body finally returns to its original condition.[20]
In this paper Clausius thought his theory would be imperfect unless it could explain non-cyclical irreversible process. So, he set about considering the interior work that he had neglected, dividing the change in the internal energy (dU) into the change in the quantity of heat contained in a system (dH) and that in the interior work (AdJ)[21].
He unified the increase in interior and exterior work into that in AdL, saying “there is no essential difference between interior and exterior work[22]," but the truth is that it makes no difference whether the interior work is classified under the internal energy or combined with the exterior work. As is evident from the equation he used, pv=R (a+t), he dealt with the ideal gas law, the combination of Mariotte’s law and Gay-Lussac’s law (Boyle’s law and Charles’s law)[23].
In those days such a gas as hydrogen, nitrogen and carbon monoxide is believed to be incapable of liquefaction and called “permanent gas." The permanent gas was known to follow the law of Mariotte and the law of Gay-Lussac with small margin of error and it corresponds to what we call the ideal gas. An ideal gas has no intermolecular forces and therefore the interior work is zero. Now that it is zero, we don’t need to care which the interior work belongs.
Clausius made a poor excuse for his theory not being applicable to the Joule expansion, saying “The law does not speak of the work which the heat does, but of the work which it can do[24]" in the paper of 1862. He made little progress in this problem in the revised version of The mechanical theory of heat in 1876[25]. One of the reasons for his failure is, as has already been explained, that his approach was thermodynamic and not statistical mechanical and he failed to recognize the increase in the equivalence value of transformations in the Joule expansion. Another is that he did not use the Van der Waals equation of real gases advocated in 1873.
The Joule expansion of a real gas, where intermolecular forces are not negligible, does the interior work in the sense of Clausius. Thanks to the law of conservation of energy, the increase in the potential energy of molecules compensates for the same amount of the decrease in their kinetic energy, namely a drop in temperature. The Joule expansion is irreversible, because the expansion increases more statistical mechanical entropy than these two factors that reduce entropy. Today we can explain the Joule expansion this way, but he could not come to this conclusion because of the two reasons.
3.2. From the equivalence value to entropy
In the paper of 1865[26], he succeeded the division of heat and work proposed in the paper of 1862 and redefined the equivalence value of transformations as follows.
Again the symbol H denotes the quantity of the heat actually present in the body whose absolute value is the only variable of this function. Clausius divided H by T to get Y and named it “the transformation-value of the heat actually present in the body[27]."
Meanwhile Clausius named Z “the disgregation of the body[28]." This concept was already introduced in the paper of 1862 to express the intermolecular distance that the interior work and the exterior work increase[29]. The concept of disgregation was later forgotten, but according to Klein, “Clausius saw the disgregation as a concept more fundamental than the entropy, since entropy was to be interpreted physically with the help of disgregation[30]."
Clausius transform Eq. 14 into
and assigned a symbol S to the sum of the transformation-value of heat and the disgregation. That is to say,
As we deal with an ideal gas, ignore the interior work and recognize their details of Y and Z. Suppose the specific heat of the gas at constant volume is c. we get the equation below[31] concerning S.
Its integration is
To adopt the current notation,
This equation indicates S increases in proportion to the logarithm of ratios of the rise in temperature and/or the volume expansion. Eq. 8 is the special case of Eq. 19, where T=T0.
What is remarkable in the paper of 1865 is that Clausius gave the name of “entropy" to the sum of the transformation-value of heat and the disgregation for the first time.
We might call S the transformation content of the body, just as we termed the magnitude U its thermal and ergonal content. But as I hold it to be better terms for important magnitudes from the ancient languages, so that they may be adopted unchanged in all modern languages, I propose to call the magnitude S the entropy of the body, from the Greek word “ἡ τροπή", transformation. I have intentionally formed the word entropy so as to be as similar as possible to the word energy; for the two magnitudes to be denoted by these words are so nearly allied their physical meanings, that a certain similarity in designation appears to be desirable.[32]
The “hē" of “hē tropē (ἡ τροπή)" is the nominative feminine singular form of the Greek article. Clausius removed it, added “en (ἐν)", a Greek preposition meaning “in" and created a new term, entropia (ἐντροπία=ἐν+τροπή) or Entropie in German. He wrote that he added “en" to assimilate entropy with energy. Does this prefix have more significance than mere assimilation?
The term “energy" derives from a Greek word “energeia (ἐνέργεια)." Unlike “entropia", it is a word that has been used since the ancient time and meant activity, operation, workmanship and so on. Aristotle used it as a metaphysical term as the antonym of “dunamis (δύναμις)"[33], of which the former is translated as actuality and the latter potentiality. It was Thomas Young (1773 – 1829) who first used this term in the modern physical sense in 1807[34]. Aristotelian usage of these terms might have contacts with the modern physical usage. When potential energy is converted into kinetic energy, we can say potentiality is turned into actuality in the Aristotelian sense.
Etymologically energeia is the combination of in (ἐν) and work (ἔργον) and literally means “in work." The “entropia" was coined in the same way. Now we can understand why Clausius wrote, “We might call S the transformation content (Verwandlungsinhalt) of the body, just as we termed the magnitude U its thermal and ergonal content (Wärme- und Werkinhalt)." Because of the first law of thermodynamics, thermal content is equal to ergonal content. The German word “Inhalt" is the combination of “in" and “hold (halten)." While the ergonal content is “what is held in work (Werk-in-halt)", namely en-ergy (ἐν-ἔργον), the transformation content is “what is held in transformtion (Verwandlungs-in-halt), namely en-tropy (ἐν-τροπή). In fact entropy is the literal translation of the transformation content or the equivalence value of transformations into Greek.
He used S as the symbol that stands for entropy in the paper of 1865 and this custom continues today, but the reason is unknown, because he did not explain it. S might be the capital letter of Sadi Carnot or the Sum (Summe in German)of Y and Z or the integral sign which is based on the Latin long S ‘’ſumma’’. Anyway entropy is the sum.
The bottom line of the paper in 1865 was the following famous formulation of the Joule law and the Carnot principle in terms of energy and entropy.
1) The energy of the world is constant.
2) The entropy of the world tends to a maximum.[35]
This is what is now called the first and the second laws of thermodynamics.
3.3. Entropy and ecological problems
Clausius stopped using the concept of disgregation in the revised version of The mechanical theory of heat in 1876. Some historians of science, however, estimate this concept. For example, Yamamoto Yoshitaka paid attention to Clausius’s distinction between Y and Z and invoked the criticism for recycling by Nicholas Georgescu-Roegen (1906 – 1994) who thought the increase in Y (the loss of energy availability) could not completely decrease Z (the loss of matter availability)[36].
To be sure, Clausius distinguished between Y (thermal diffusion) and Z (matter mixing), but he did not think that we cannot transform the latter entropy into the former entropy. As the original name of the equivalence value of transformations suggests, he regarded two transoformations from Z to Y and from Y to Z as equivalent.
The disgregation depends on the arrangement of the particles of the body, and the measure of an increment of disgregation is the equivalence-value of the transformation from ergon to heat which must take place in order to cancel the increment of disgregation, and thus serve as a substitute for that increment.[37]
This quotation from the paper of 1865 shows that Clausius thought the transformation from ergon (work) to heat can decrease disgregation. The exchange is depicted in the figure below.

In fact recycling, a decrease in ΔZ by means of an increase in ΔY, is theoretically possible. Of course, the Earth as a heat engine has its limit of capacity for recycling, but it is the matter of quantity and not the matter of principle. We can transform the entropy of matter diffusion into the entropy of thermal diffusion, throw away the heat to the outer space and run a sustainable economy so long as we do not exceed the limit.
Clausius’s conclusion is not “The entropy of the Earth tends to a maximum" but “The entropy of the world tends to a maximum." The second law of thermodynamics can apply only to an isolated system. It does not necessarily apply to a non-isolated system like the Earth. The Earth is a heat engine whose hot reservoir of heat is solar radiation and geothermal heat and whose cold reservoir of heat is outer space.
The average heat flux density of solar radiation per square meter that the Earth receives is 341.5 W/m2. Clouds reflect some of the radiation and the surface of the Earth receives approximately 70% of it, 239 W/m2. The temperature of the surface of the Earth is 15℃ (288K) and that of the tropopause (the boundary between the troposphere and the stratosphere) is -24℃ (249K) so that solar radiation reduces entropy by
The average heat flux density from Earth’s interior to its crust per square meter is 91.6 mW/m2. The temperature of the core–mantle boundary is about 4000K so that geothermal heat reduces entropy by
As geothermal heat is also thrown away into outer space, the total entropy it reduces amounts to
Owing to these reductions, the Earth does not become a dead planet unless we increase more entropy than the Earth as a heat engine can decrease.
4. References
- ↑“Bei näherer Betrachtung findet man aber, dass nicht das eigentliche Grundprincip von Carnot, sondern nur der Zusatz, dass keine Wärme verloren gehe, der neuen Betrachtungsweise entgegensteht, denn es kann bei der Erzeugung von Arbeit sehr wohl beides gleichzeitig stattfinden, dass eine gewisse Wärmemenge verbraucht und eine andere von einem warmen zu einem kalten Körper übergeführt wird, und beide Wärmemengen können zu der erzeugten Arbeit in bestimmter Beziehung stehen." Clausius, Rudolf Julius Emmanuel. “Über Die Bewegende Kraft Der Wärme Und Die Gesetze, Welche Sich Daraus Für Die Wärmelehre Selbst Ableiten Lassen.” First published: Annalen Der Physik 155, no. 3 (n.d.): 368–97. 1850. Reprinted: Abhandlungen Über Die Mechanische Wärmetheorie. p.20.
- ↑“La production de la puissance motrice est donc due, dans les machines à vapeur, non à une consommation réelle du calorique, mais à son transport d’un corps chaud à un corps froid” Nicolas Léonard Sadi Carnot. Réflexions sur la puissance motrice du feu et sur les machines propres à développer cette puissance. First published: 1824. p. 10-11.
- ↑Carnot, Nicolas Léonard Sadi. “Extrait de notes inédites de Sadi Carnot” in Réflexions sur la puissance motrice du feu et sur les machines propres à développer cette puissance. First published: 1824. p. 92.
- ↑“It is impossible, by means of inanimate material agency, to derive mechanical effect from any portion of matter by cooling it below the temperature of the coldest of the surrounding objects." Thomson, W. (1851). “On the Dynamical Theory of Heat, with numerical results deduced from Mr Joule’s equivalent of a Thermal Unit, and M. Regnault’s Observations on Steam". Transactions of the Royal Society of Edinburgh. XX (part II): §12.
- ↑“Da nämlich auch ein Wärmeübergang ohne mechanischen Effect stattfinden kann, wenn ein warmer und ein kalter Körper sich unmittelbar berühren, und die Wärme durch Leitung hinüberströmt, so muss, wenn man für den Übergang einer bestimmten Wärmemenge zwischen zwei Körpern von bestimmten Temperaturen t und τ das Maximum der Arbeit erlangen will, der Vorgang so geleitet werden, wie es in den obigen Fällen geschehen ist, dass nie zwei Körper von verschiedener Temperatur in Berührung kommen." Clausius, Rudolf Julius Emmanuel (1850). “Über Die Bewegende Kraft Der Wärme Und Die Gesetze, Welche Sich Daraus Für Die Wärmelehre Selbst Ableiten Lassen.” in Abhandlungen Über Die Mechanische Wärmetheorie. p.49.
- ↑Rudolf Clausius. “Über die bewegende Kraft der Wärme: und die Gesetze, welche sich daraus für die Wärmelehre selbst ableiten lassen." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 1. 1864. p. 32.
- ↑“die hinzugekommene freie Wärme und die zu innerer Arbeit, falls solche geschehen ist, verbrauchte Wärme umfasst" Rudolf Clausius. “Über die bewegende Kraft der Wärme: und die Gesetze, welche sich daraus für die Wärmelehre selbst ableiten lassen." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 1. 1864. p. 33.
- ↑“Die geleistete Arbeit ist nämlich von zweifacher Art. Erstens gehört eine gewisse Arbeit dazu, um die gegenseitige Anziehungskraft der Theilchen des Wassers zu überwinden, und sie bis zu der Entfernung von einander zu trennen, in welcher sie sich beim Dampfe befinden. Zweitens muss der Dampf bei seiner Entwickelung einen äusseren Druck zurückschieben, um sich Raum zu schaffen. Die erstere Arbeit wollen wir die innere, die letztere die äussere nennen, und danach auch die latente Wärme eintheilen." Rudolf Clausius. “Über die bewegende Kraft der Wärme: und die Gesetze, welche sich daraus für die Wärmelehre selbst ableiten lassen." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 1. 1864. p. 23.
- ↑“Wenn irgend ein Körper sein Volumen verändert, so wird dabei im Allgemeinen mechanische Arbeit erzeugt oder verbraucht. Es ist aber in den meisten Fällen nicht möglich, diese genau zu bestimmen, weil zugleich mit der äusseren Arbeit auch gewöhnlich noch eine unbekannte innere stattfindet. Um diese Schwierigkeit zu vermeiden, hat Carnot das schon oben erwähnte sinnreiche Verfahren angewandt, dass er den Körper nach einander verschiedene Veränderungen durchmachen lässt, die so angeordnet sind, dass er zuletzt wieder genau in seinen ursprünglichen Zustand zurückkommt." Rudolf Clausius. “Über die bewegende Kraft der Wärme: und die Gesetze, welche sich daraus für die Wärmelehre selbst ableiten lassen." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 1. 1864. p. 26.
- ↑“熱を全て仕事にすることはできない" 「ウィリアム・トムソン」 Wikipedia. 2013年10月27日.
- ↑“It is impossible, by means of inanimate material agency, to derive mechanical effect from any portion of matter by cooling it below the temperature of the coldest of the surrounding objects." William Thomson. On the Dynamical Theory of Heat. Taylor & Francis (1852).
- ↑“Diese letztere Constante ist also für die verschiedenen Gase in sofern verschieden, als sie ihrem specifischen Gewichte umgekehrt proportional ist." Rudolf Clausius. “Über die bewegende Kraft der Wärme: und die Gesetze, welche sich daraus für die Wärmelehre selbst ableiten lassen." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 1. 1864. p. 24-25.
- ↑Rudolf Clausius. “Über die bewegende Kraft der Wärme: und die Gesetze, welche sich daraus für die Wärmelehre selbst ableiten lassen." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 1. 1864. p. 47.
- ↑“Lorsqu’un gaz varie de volume sans changer de température, les quantités de chaleur absorbées ou dégagées par ce gaz sont en progression arithmétique, si les accroissemens ou les réductions de volume se trouvent être en progression géométrique" Nicolas Léonard Sadi Carnot. Réflexions sur la puissance motrice du feu et sur les machines propres à développer cette puissance. First published: 1824. p. 52-53.
- ↑“In allen Fällen, wo eine Wärmemenge in Arbeit verwandelt wird, und der diese Verwandlung vermittelnde Körper sich schliesslich wieder in seinem Anfangszustande befindet, muss zugleich eine andere Wärmemenge aus einem wärmeren in einen kälteren Körper übergehen, und die Grösse der letzteren Wärmemenge im Verhältniss zur ersteren ist nur von den Temperaturen der beiden Körper, zwischen welchen sie übergeht, und nicht von der Art des vermittelnden Körpers abhängig." Rudolf Clausius. “Über eine veränderte Form des zweiten Hauptsatzes der mechanischen Wärmetheorie." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 1. 1864. p. 133.
- ↑“es kann nie Wärme aus einem kälteren in einen wärmeren Kärper übergehen, wenn nicht gleichzeitig eine andere damit zusam/nienhängende Aenderung eintritt" Rudolf Clausius. “Über eine veränderte Form des zweiten Hauptsatzes der mechanischen Wärmetheorie." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 1. 1864. p. 134.
- ↑“Aequivalenzwerth der Verwandlungen" Rudolf Clausius. “Über eine veränderte Form des zweiten Hauptsatzes der mechanischen Wärmetheorie." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 1. 1864. p. 143.
- ↑“eine uncompensirte Verwandlungen" Rudolf Clausius. “Über eine veränderte Form des zweiten Hauptsatzes der mechanischen Wärmetheorie." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 1. 1864. p. 152.
- ↑“auf die wirkliche Ausführung für einzelne specielle Fälle will ich hier nicht eingehen" Rudolf Clausius. “Über eine veränderte Form des zweiten Hauptsatzes der mechanischen Wärmetheorie." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 1. 1864. p. 152.
- ↑“Da ich nun in meiner früheren Veröffentlichung alles Hypothetische zu veriueiden wünschte, so schloss ich die innere Arbeit ganz davon aus, was dadurch geschehen konnte, dass ich mich auf die Betrachtung von Kreisprocessen beschränkte, d. h. von Vorgängen, bei denen die Veränderungen, welche der Körper erleidet, so angeordnet sind, dass der Körper schliesslich wieder in seinen Anfangszustand zurückkommt." Rudolf Clausius. “Über die Anwendung des Satzes von der Aequivalenz der Verwandlungen auf die innere Arbeit." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 1. 1864. p. 243.
- ↑Rudolf Clausius. “Über die Anwendung des Satzes von der Aequivalenz der Verwandlungen auf die innere Arbeit." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 1. 1864. p. 253.
- ↑“die innere und äussere Arbeit nicht wesentlich von einander verschieden sind" Rudolf Clausius. “Über die Anwendung des Satzes von der Aequivalenz der Verwandlungen auf die innere Arbeit." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 1. 1864. p. 246.
- ↑Rudolf Clausius. “Über die bewegende Kraft der Wärme: und die Gesetze, welche sich daraus für die Wärmelehre selbst ableiten lassen." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 1. 1864. p. 24.
- ↑“Das Gesetz spricht nicht von der Arbeit, welche die Wärme thut, sondern von der, welche sie thun kann" Rudolf Clausius. “Über die Anwendung des Satzes von der Aequivalenz der Verwandlungen auf die innere Arbeit." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 1. 1864. p. 251.
- ↑Rudolf Clausius. Die mechanische wärmetheorie. 1876. p. 223-228.
- ↑Clausius, Rudolf. Über verschiedene für die Anwendung bequeme Formen der Hauptgleichungen der mechanischen Wärmetheorie. In: Annalen der Physik und Chemie. Band 125. Barth Leipzig (1865). Vorgetragen in der naturforsch. Gesellschaft den 24. April 1865.
- ↑“den Verwandlungswerth der im Körper vorhandenen Wärme" Rudolf Clausius. “Über verschiedene für die Anwendung bequeme Formen der Hauptgleichungen der mechanischen Wärmetheorie." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 2. 1867. p. 32.
- ↑“die Disgregation des Körpers" Rudolf Clausius. “Über verschiedene für die Anwendung bequeme Formen der Hauptgleichungen der mechanischen Wärmetheorie." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 2. 1867. p. 33.
- ↑Rudolf Clausius. “Über die Anwendung des Satzes von der Aequivalenz der Verwandlungen auf die innere Arbeit." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 1. 1864. p. 248.
- ↑Martin J. Klein. “Gibbs on Clausius." Historical Studies in the Physical Sciences 1 (1969): 127-149.
- ↑Rudolf Clausius. “Über verschiedene für die Anwendung bequeme Formen der Hauptgleichungen der mechanischen Wärmetheorie." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 2. 1867. p. 37.
- ↑“Sucht man für S einen bezeichnenden Namen, so könnte man, ähnlich wie von der Grösse U gesagt ist, sie sei der Wärme- und Werkinhalt des Körpers, von der Grösse S sagen , sie sei der Verwandlungsinhalt des Körpers. Da ich es aber für besser halte, die Namen derartiger für die Wissenschaft wichtiger Grössen aus den alten Sprachen zu entnehmen, damit sie unverändert in allen neuen Sprachen angewandt werden können, so schlage ich vor, die Grösse S nach dem griechischen Worte ἡ τροπή, die Verwandlung, die Entropie des Körpers zu nennen. Das Wort Entropie habe ich absichtlich dem Worte Energie möglichst ähnlich gebildet, denn die beiden Grossen, welche durch diese Worte benannt werden sollen, sind ihren physikalischen Bedeutungen nach einander so nahe verwandt, dass eine gewisse Gleichartigkeit in der Benennung mir zweckmässig zu sein scheint." Rudolf Clausius. “Über verschiedene für die Anwendung bequeme Formen der Hauptgleichungen der mechanischen Wärmetheorie." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 2. 1867. p. 34.
- ↑Aristotle. Metaphysics.Book 9, section 1047a.
- ↑Thomas Young. A course of lectures on natural philosophy and the mechanical arts. Lecture 8. p. 78.
- ↑“Die Energie der Welt ist constant. Die Entropie der Welt strebt einem Maximum zu." Rudolf Clausius. “Über verschiedene für die Anwendung bequeme Formen der Hauptgleichungen der mechanischen Wärmetheorie." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 2. 1867. p. 44.
- ↑“実際、大気中への窒素化合物や硫黄化合物の拡散、水銀やカドミウム等重金属類の土中や海中への拡散は、すべて非可逆的なエントロピーの増大、クラウジウスの言う〈分散Z〉の増大であって、それを人工的に元に戻そうとすれば、おびただしいエネルギーの消費としたがってまたさらなるエントロピーの発生をもたらすことになる。[…]この意味で、人類の生存条件の維持にとっては、熱拡散に劣らず物拡散の防止がより重要な要因になる。YとZの和としてエントロピーを考えるクラウジウスの出発点の現代的重要性はここにある。" 山本義隆. 『熱学思想の史的展開』現代数学社 (January 1, 1987). p. 481-482.
- ↑“Sie [die Disgregation des Körpers] hängt von der Anordnung der Bestandtheile des Körpers ab, und das Maass einer Disgregationsvermehrung ist der Aequivalenzwerth derjenigen Verwandlung aus Werk in Wärme, welche stattfinden muss, um die Disgregationsvermehrung wieder rückgängig zu machen, welche also als Ersatz der Disgregationsvermehrung dienen kann." Rudolf Clausius. “Über verschiedene für die Anwendung bequeme Formen der Hauptgleichungen der mechanischen Wärmetheorie." In: Abhandlungen Über Die Mechanische Wärmetheorie. Vol. 2. 1867. p. 33.








Discussion
New Comments
hi this is teja
your article is very nice.
can you explain this line ” it was still unknown in those days where the heat is gone that is not converted to work and the Carnot cycle which has no difficulty in explaining this problem could simplify the model of thermodynamics”
im bit confused on what part of carnot cycle your are taking about
thanks in adavance.
The sentence you pointed out is certainly misleading. So, I have revised it as follows.
I mean the Carnot cycle holds true whether it presumes the conservation of heat or not.
i can understand all your concept but i was not able to join together all of them to understand what entropu is
Exacctly i want to say is
1)how carnot cycle lead clausius to define equivalance value of tranformation?
2)what is the significance of equivalance value of transformation in efficiency of heat engine?
and many doubts which are not satisfing in understanding entropy.
i hope you have free time to discuss about his topic, if not whenever your free please intimate me on email address.
thanks in advance.
Imagine you were a person who invented money. You made somebody work for you and gave him or her a reward. You would find something valuable in common between the two things. It is no wonder that you should come up with the idea of money that stands for the same value.
That would be the same process that Clausius came up with the idea of the equivalence value of transformations or entropy. Entropy means the negative value. Here it is not important whether the new concept of value is positive or negative. If you spend money, you get a negative value. If you earn money, you get a positive money. It is just a relative matter.
The Carnot cycle transforms reducing the temperature difference into a useful work. Reducing the temperature difference reduces value, namely increases the equivalence value of transformations or entropy that a useful work can compensate for.
When you spend money, you would like to get the maximum value at the cost of the minimum money. The same efforts are made to improve the efficiency of the heat engine. Engineers have tried to improve the heat engines so that they can extract the maximum useful work at the minimum entropy.
Social media such as Facebook or twitter would be a better way for that purpose. If you want to contact me in private, feel free to use the mail form and send your message.